Describe the Process of Making Babys Describe the Process of Osmosis

The process of osmosis over a semi-permeable membrane. The blueish dots represent particles driving the osmotic gradient.
Osmosis (, )[1] is the spontaneous net motion or diffusion of solvent molecules through a selectively permeable membrane from a region of high water potential (region of lower solute concentration) to a region of depression h2o potential (region of higher solute concentration),[2] in the direction that tends to equalize the solute concentrations on the 2 sides.[iii] [four] [5] It may also be used to describe a concrete procedure in which whatsoever solvent moves across a selectively permeable membrane (permeable to the solvent, but non the solute) separating two solutions of unlike concentrations.[6] [vii] Osmosis tin can be made to do work.[8] Osmotic pressure is defined every bit the external pressure required to be applied and then that there is no cyberspace movement of solvent across the membrane. Osmotic pressure is a colligative property, significant that the osmotic pressure level depends on the tooth concentration of the solute but not on its identity.
Osmosis is a vital process in biological systems, as biological membranes are semipermeable. In general, these membranes are impermeable to large and polar molecules, such as ions, proteins, and polysaccharides, while being permeable to not-polar or hydrophobic molecules like lipids besides every bit to small molecules like oxygen, carbon dioxide, nitrogen, and nitric oxide. Permeability depends on solubility, charge, or chemistry, as well equally solute size. H2o molecules travel through the plasma membrane, tonoplast membrane (vacuole) or protoplast past diffusing beyond the phospholipid bilayer via aquaporins (small transmembrane proteins like to those responsible for facilitated diffusion and ion channels). Osmosis provides the primary means by which water is transported into and out of cells. The turgor pressure level of a cell is largely maintained past osmosis across the jail cell membrane between the cell interior and its relatively hypotonic environment.
History

Some kinds of osmotic flow take been observed since aboriginal times, e.g., on the construction of Egyptian pyramids.[9] Jean-Antoine Nollet start documented observation of osmosis in 1748.[10] The give-and-take "osmosis" descends from the words "endosmose" and "exosmose", which were coined past French physician René Joachim Henri Dutrochet (1776–1847) from the Greek words ἔνδον (éndon "inside"), ἔξω (éxō "outer, external"), and ὠσμός (ōsmós "push, impulsion").[xi] In 1867, Moritz Traube invented highly selective precipitation membranes, advancing the art and technique of measurement of osmotic flow.[nine]
Description
Osmosis is the motion of a solvent across a semipermeable membrane toward a higher concentration of solute. In biological systems, the solvent is typically water, but osmosis can occur in other liquids, supercritical liquids, and even gases.[12] [13]
When a prison cell is submerged in water, the water molecules pass through the cell membrane from an area of low solute concentration to high solute concentration. For example, if the prison cell is submerged in saltwater, water molecules move out of the cell. If a cell is submerged in freshwater, h2o molecules movement into the prison cell.

Water passing through a semi-permeable membrane
When the membrane has a volume of pure water on both sides, water molecules pass in and out in each direction at exactly the same rate. There is no net flow of h2o through the membrane.
Osmosis tin be demonstrated when potato slices are added to a high salt solution. The water from inside the spud moves out to the solution, causing the potato to shrink and to lose its 'turgor pressure'. The more than full-bodied the salt solution, the bigger the loss in size and weight of the potato slice.
Chemical gardens demonstrate the event of osmosis in inorganic chemical science.
Machinery
The mechanism responsible for driving osmosis has unremarkably been represented in biology and chemical science texts equally either the dilution of water past solute (resulting in lower concentration of water on the higher solute concentration side of the membrane and therefore a diffusion of h2o along a concentration gradient) or by a solute's attraction to water (resulting in less free water on the higher solute concentration side of the membrane and therefore cyberspace movement of water toward the solute). Both of these notions have been conclusively refuted.
The improvidence model of osmosis is rendered untenable by the fact that osmosis can drive h2o across a membrane toward a higher concentration of water.[xiv] The "bound water" model is refuted by the fact that osmosis is independent of the size of the solute molecules—a colligative property[15]—or how hydrophilic they are.
It is difficult to describe osmosis without a mechanical or thermodynamic caption, but substantially there is an interaction betwixt the solute and water that counteracts the pressure that otherwise gratis solute molecules would exert. One fact to accept note of is that heat from the environs is able to be converted into mechanical free energy (water rising).
Many thermodynamic explanations go into the concept of chemical potential and how the part of the h2o on the solution side differs from that of pure water due to the higher pressure and the presence of the solute counteracting such that the chemical potential remains unchanged. The virial theorem demonstrates that allure between the molecules (water and solute) reduces the force per unit area, and thus the pressure exerted by water molecules on each other in solution is less than in pure water, allowing pure h2o to "forcefulness" the solution until the force per unit area reaches equilibrium.[15]
Office in living things

Issue of different solutions on blood cells

Micrographs of osmotic force per unit area on red blood cells (RBC)
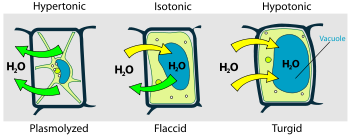
Plant cell in unlike environments.
Osmotic pressure is the main amanuensis of back up in many plants. The osmotic entry of h2o raises the turgor pressure exerted against the cell wall, until information technology equals the osmotic pressure, creating a steady state.
When a plant cell is placed in a solution that is hypertonic relative to the cytoplasm, water moves out of the cell and the cell shrinks. In doing so, the prison cell becomes flaccid. In extreme cases, the jail cell becomes plasmolyzed – the prison cell membrane disengages with the prison cell wall due to lack of water pressure on information technology.
When a plant cell is placed in a solution that is hypotonic relative to the cytoplasm, water moves into the cell and the cell swells to go turgid.
Osmosis is responsible for the ability of plant roots to describe water from the soil. Plants concentrate solutes in their root cells by agile send, and h2o enters the roots past osmosis. Osmosis is too responsible for controlling the movement of guard cells.
In unusual environments, osmosis tin be very harmful to organisms. For example, freshwater and saltwater aquarium fish placed in water of a different salinity than that to which they are adapted to volition die quickly, and in the case of saltwater fish, dramatically. Some other example of a harmful osmotic effect is the use of table common salt to kill leeches and slugs.
Suppose an animal or a plant cell is placed in a solution of carbohydrate or common salt in h2o.
- If the medium is hypotonic relative to the cell cytoplasm, the cell will gain water through osmosis.
- If the medium is isotonic, there volition be no net movement of water across the prison cell membrane.
- If the medium is hypertonic relative to the prison cell cytoplasm, the cell volition lose water by osmosis.
This ways that if a cell is put in a solution which has a solute concentration higher than its ain, information technology will shrivel, and if information technology is put in a solution with a lower solute concentration than its ain, the cell will bully and may fifty-fifty flare-up.
Factors
Osmotic pressure level
Osmosis may be opposed past increasing the pressure level in the region of high solute concentration with respect to that in the depression solute concentration region. The strength per unit of measurement surface area, or pressure level, required to prevent the passage of water (or any other high-liquidity solution) through a selectively permeable membrane and into a solution of greater concentration is equivalent to the osmotic force per unit area of the solution, or turgor. Osmotic pressure is a colligative belongings, meaning that the property depends on the concentration of the solute, but non on its content or chemic identity.
Osmotic gradient
The osmotic gradient is the difference in concentration between two solutions on either side of a semipermeable membrane, and is used to tell the deviation in percentages of the concentration of a specific particle dissolved in a solution.
Usually the osmotic slope is used while comparison solutions that have a semipermeable membrane between them allowing water to diffuse between the ii solutions, toward the hypertonic solution (the solution with the higher concentration). Eventually, the force of the column of water on the hypertonic side of the semipermeable membrane will equal the forcefulness of diffusion on the hypotonic (the side with a lesser concentration) side, creating equilibrium. When equilibrium is reached, water continues to menstruum, but it flows both ways in equal amounts also as strength, therefore stabilizing the solution.
Variation
Reverse osmosis
Opposite osmosis is a separation process that uses pressure level to force a solvent through a semi-permeable membrane that retains the solute on ane side and allows the pure solvent to laissez passer to the other side, forcing information technology from a region of high solute concentration through a membrane to a region of depression solute concentration by applying a pressure in excess of the osmotic force per unit area.
Forrard osmosis
Osmosis may be used directly to reach separation of water from a solution containing unwanted solutes. A "depict" solution of higher osmotic force per unit area than the feed solution is used to induce a net menstruation of water through a semi-permeable membrane, such that the feed solution becomes full-bodied as the draw solution becomes dilute. The diluted draw solution may so be used direct (as with an ingestible solute similar glucose), or sent to a secondary separation process for the removal of the draw solute. This secondary separation can be more than efficient than a contrary osmosis process would be alone, depending on the depict solute used and the feedwater treated. Forrad osmosis is an area of ongoing inquiry, focusing on applications in desalination, water purification, water treatment, nutrient processing, and other areas of study.
Meet as well
- Brining
- Homeostasis
- Osmoregulation
- Osmotic stupor
- Osmotic ability
- Plasmolysis
- Opposite osmosis constitute
- Salinity gradient power
- H2o potential
References
- ^ Jones, Daniel (2011). Roach, Peter; Setter, Jane; Esling, John (eds.). Cambridge English language Pronouncing Dictionary (18th ed.). Cambridge University Printing. ISBN978-0-521-15255-half-dozen.
- ^ "Osmosis | A Level Notes".
{{cite spider web}}: CS1 maint: url-condition (link) - ^ "Osmosis". Oxford English Dictionary (Online ed.). Oxford Academy Printing. (Subscription or participating institution membership required.)
- ^ Osmosis, Encyclopædia Britannica on-line
- ^ Haynie, Donald T. (2001). Biological Thermodynamics . Cambridge: Cambridge Academy Press. pp. 130–136. ISBN978-0-521-79549-4.
- ^ Waugh, A.; Grant, A. (2007). Anatomy and Physiology in Health and Illness. Edinburgh: Elsevier. pp. 25–26. ISBN978-0-443-10101-4.
- ^ Osmosis Archived 22 February 2008 at the Wayback Automobile. University of Hamburg. last change: 31 July 2003
- ^ "Statkraft to build the world's showtime prototype osmotic ability found". Statkraft. 3 October 2007. Archived from the original on 27 Feb 2009.
- ^ a b Hammel, H.T.; Scholander, P.F. (1976). Perspectives on the Machinery of Osmosis and Imbibition In: Osmosis and tensile solvent. Springer-Verlag, Berlin, Heidelberg, New York. link.
- ^ Fifty'Abbé Nollet (June 1748) "Recherches sur les causes du bouillonnement des liquides" (Researches on the causes of the humid of liquids) Mémoires de Mathématique et de Physique, tirés des registres de l'Académie Royale des Sciences de l'année 1748, pp. 57–104; especially pp. 101–103. The Mémoires (1748) were printed in: Histoire de l'Académie Royale des Sciences Année 1748, which was published in 1752 and which contains a condensed version of Nollet'due south article on pages 10–19.
Original text : Avant que de finir ce Mémoire, je crois devoir rendre compte d'un fait que je dois au hasard, & qui me parut d'abord … singulier … j'en avois rempli une fiole cylindrique, longue de cinq pouces, & d'un pouce de diamètre ou environ ; & l'ayant couverte d'united nations morceau de vessie mouillée & ficelée au col du vaisseau, je l'avois plongée dans un grand vase plein d'eau, afin d'être sûr qu'il ne rentrât aucun air dans l'camaraderie de vin. Au bout de cinq ou six heures, je fus tout surpris de voir que la fiole étoit plus pleine qu'au moment de son immersion, quoiqu'elle le fût alors autant que ses bords pouvoient le permettre ; la vessie qui lui servoit de bouchon, étoit devenue convexe & si tendue, qu'en la piquant avec une épingle, il en sortit united nations jet de liqueur qui s'éleva à plus d'united nations pied de hauteur.
Translation : Before finishing this memoir, I call up I should report an event that I owe to chance and which at start seemed to me … strange … I filled [with alcohol] a cylindrical vial, five inches long and about one inch in bore; and [after] having covered information technology with piece of damp bladder [which was] tied to the neck of the vial, I immersed information technology in a large bowl full of water, in society to be sure that no air re-entered the booze. At the end of 5 or 6 hours, I was very surprised to see that the vial was fuller than at the moment of its immersion, although it [had been filled] as far as its sides would allow ; the float that served equally its cap, bulged and had become so stretched that on pricking it with a needle, in that location came from it a jet of alcohol that rose more than than a pes loftier.
- ^ Etymology of "osmosis" :
- Henri Dutrochet, 50'Amanuensis Immédiat du Motility Vital Dévoilé dans sa Nature et dans son Way d'Activeness chez les Végétaux et chez les Animaux [The firsthand agent of living movement, its nature and mode of action revealed in plants and animals] (Paris, France: Dentu, 1826), pp. 115 and 126.
- The intermediate discussion "osmose" and the word "osmotic" were coined past Scottish chemist Thomas Graham. Come across: Thomas Graham (1854) "VII. The Bakerian Lecture – On Osmotic Strength," Philosophical Transactions of the Royal Club (London), vol. 144, pp. 177–288; encounter peculiarly pp. 177, 178, and 227. See also: Thomas Graham and Henry Watts, Elements of Chemistry: Including the Applications of the Sciences in the Arts, 2nd ed. (London, England: Hippolyte Bailliere, 1858), vol. 2, p. 616.
- The word "osmosis" first appeared in: Jabez Hogg, The Microscope: Its History, Structure, and Application..., 6th ed. (London, England: George Routledge and Sons, 1867), p. 226.
- The etymology of the word "osmosis" is discussed in: Homer W. Smith (1960). "I. Theory of Solutions: A knowledge of the laws of solutions". Circulation. 21 (v): 808–817 (810). doi:ten.1161/01.CIR.21.5.808. PMID 13831991.
- ^ Kramer, Eric; David Myers (2013). "Osmosis is not driven by h2o dilution". Trends in Institute Science. 18 (iv): 195–197. doi:ten.1016/j.tplants.2012.12.001. PMID 23298880.
- ^ Kramer, Eric; David Myers (2012). "5 pop misconceptions of osmosis". American Journal of Physics. eighty (694): 694–699. Bibcode:2012AmJPh..80..694K. doi:10.1119/1.4722325.
- ^ Kosinski, R. J.; C. K. Morlok (2008). "Challenging misconceptions about osmosis". Association for Biological science Laboratory Teaching. 30: 63–87.
- ^ a b Borg, Frank (2003). "What is osmosis? Caption and understanding of a physical phenomenon". arXiv:physics/0305011.
External links
| | Look upwards osmosis in Wiktionary, the complimentary dictionary. |
| | Wikimedia Commons has media related to Osmosis. |
- Osmosis simulation in Java
- NetLogo Osmosis simulation for educational utilise
- An Osmosis Experiment
crockerbeader1974.blogspot.com
Source: https://en.wikipedia.org/wiki/Osmosis
0 Response to "Describe the Process of Making Babys Describe the Process of Osmosis"
ارسال یک نظر